He literally just nutted in me and started shopping for fine pentaerythritol, 70% AR grade nitric acid,98% Sulfuric acid, acetone, filter paper, 300 ml beakers, a thermometer, distilled water, and baking soda. - )

Question Video: Determining the Concentration of Sulfuric Acid Via Titration with Sodium Carbonate | Nagwa
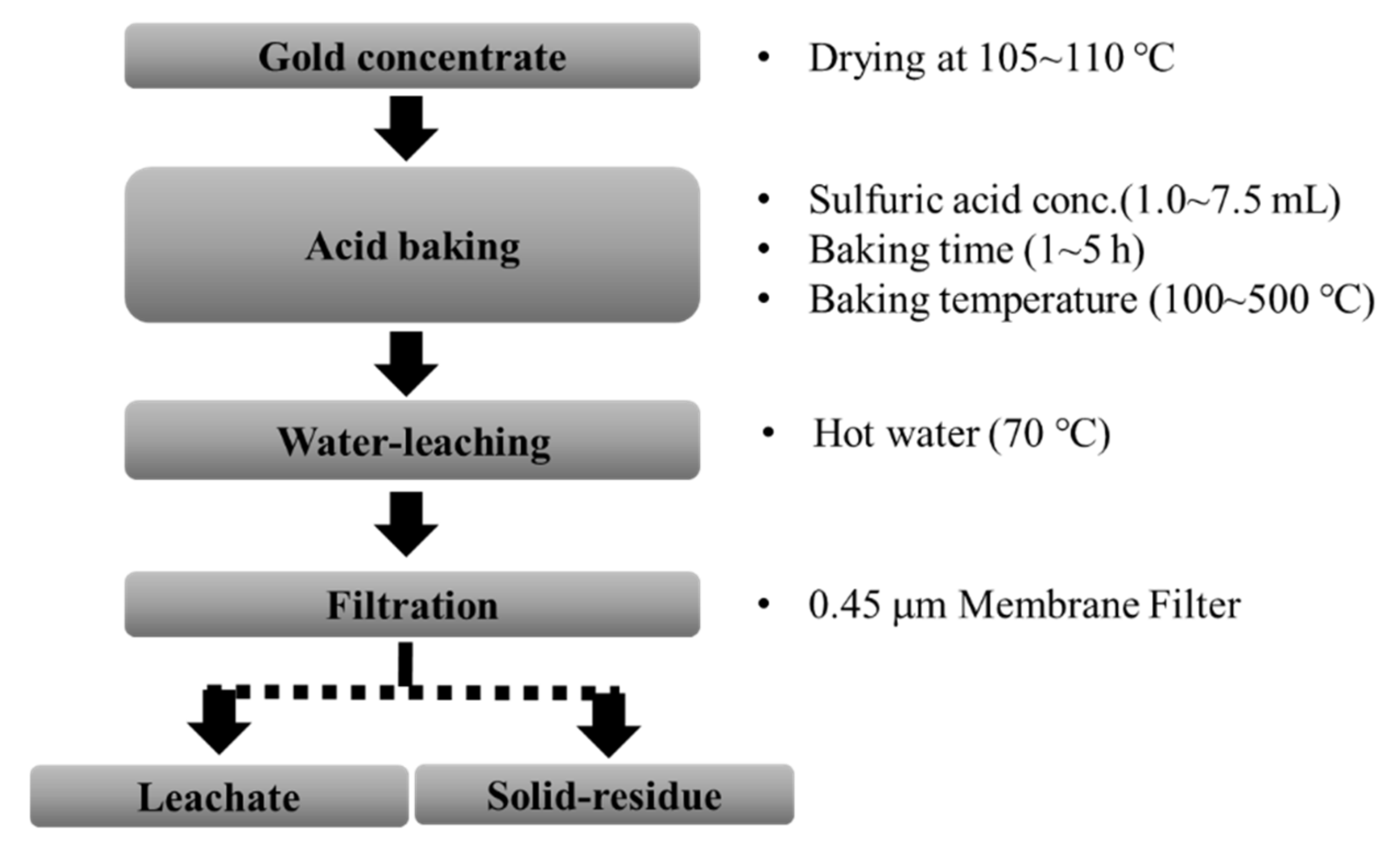
Minerals | Free Full-Text | Sulfuric Acid Baking—Water Leaching for Gold Enrichment and Arsenic Removal from Gold Concentrate | HTML

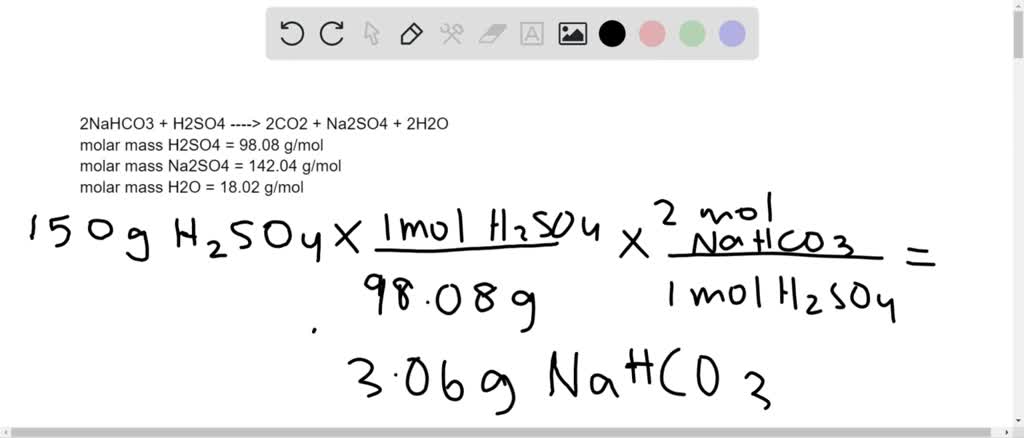
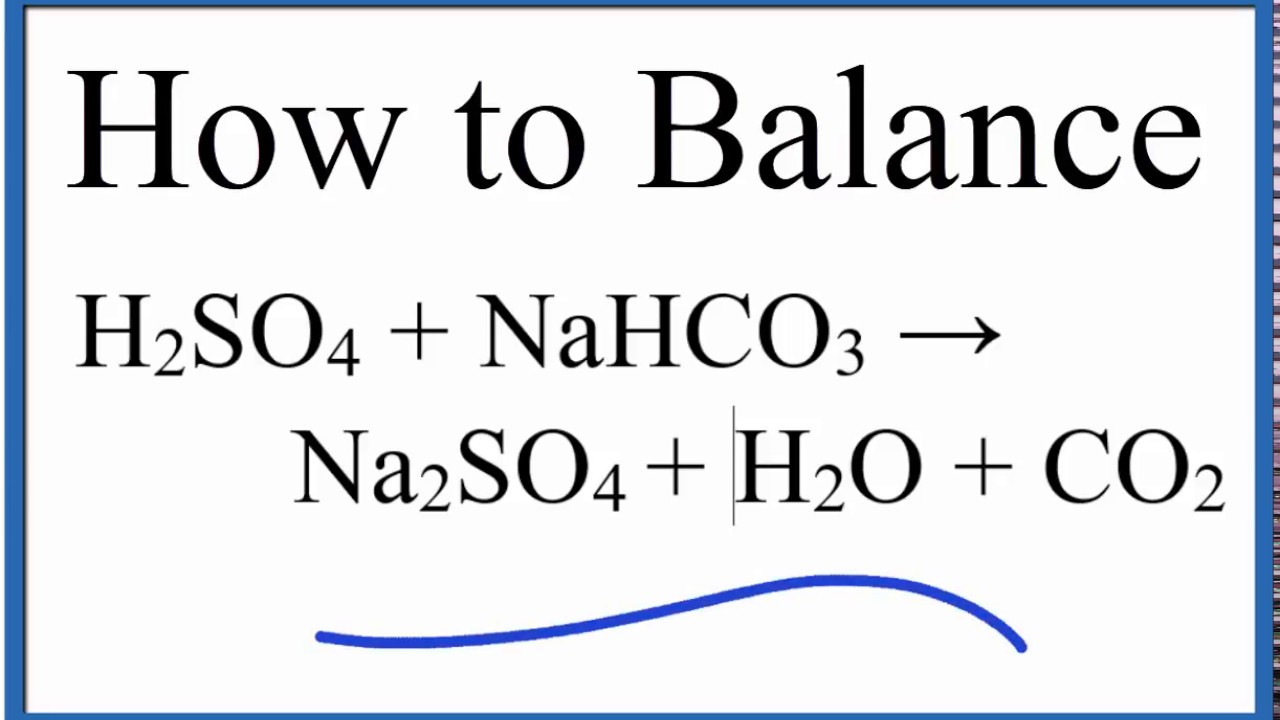
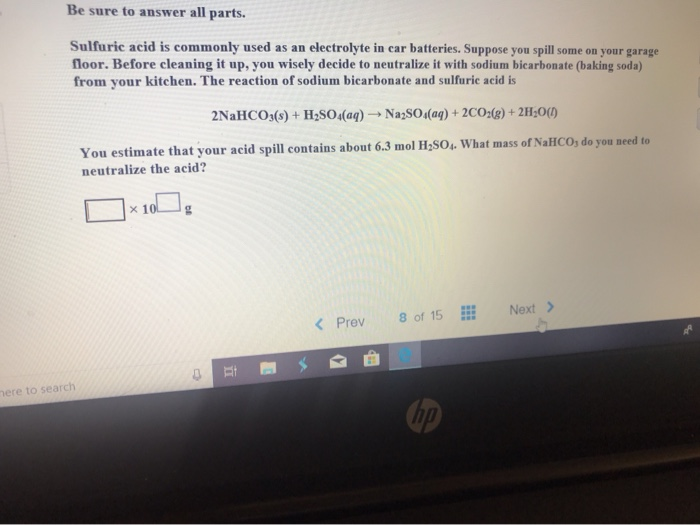
SOLVED: Aqueous solutions of sodium bicarbonate and sulfuric acid react to produce carbon dioxide according to the following equation: 2 NaHCOs(aq) + H,SO4(aq) 3 2CO2(g) + NazSO4(aq) + 2 HzO() If 105.4

SOLVED: Aqueous solutions of sodium bicarbonate and sulfuric acid react to produce carbon dioxide according to the following equation: 2 NaHCO3(aq) + H,SO4(aq) = 2 C02(g) + Na2 SO4(aq) +2H20(1) If 106.6